Table of Contents
KanBo – The Pharma-Focused Work Coordination Maestro
Experience ultimate task alignment, communication and collaboration
Trusted globally, KanBo, bridges the gap between management and engineering in complex pharmaceutical organizations. Seamless coordination, advanced project planning, and outstanding leadership are made possible through our versatile software. Stride toward your mission-critical goals with superior collaboration and communication.
KanBo for Senior Principal Biostatistician in the Pharmaceutical Industry: Boosting Collaboration, Efficiency and Quality
What do you need to know about this challenge?
A Senior Principal Biostatistician in the pharmaceutical industry plays a key role in driving the efficiency and efficacy of clinical trial teams and other quantitative partners. The challenge lays in coordinating with various teams to implement experimental designs and analyses, while ensuring timeliness and the quality of statistical deliverables. They have to maintain smooth communication flow, efficient task allocation, and effective progress tracking.
What can you do with KanBo to solve this challenge?
KanBo, with its array of features, provides a viable solution to these collaboration and management hurdles:
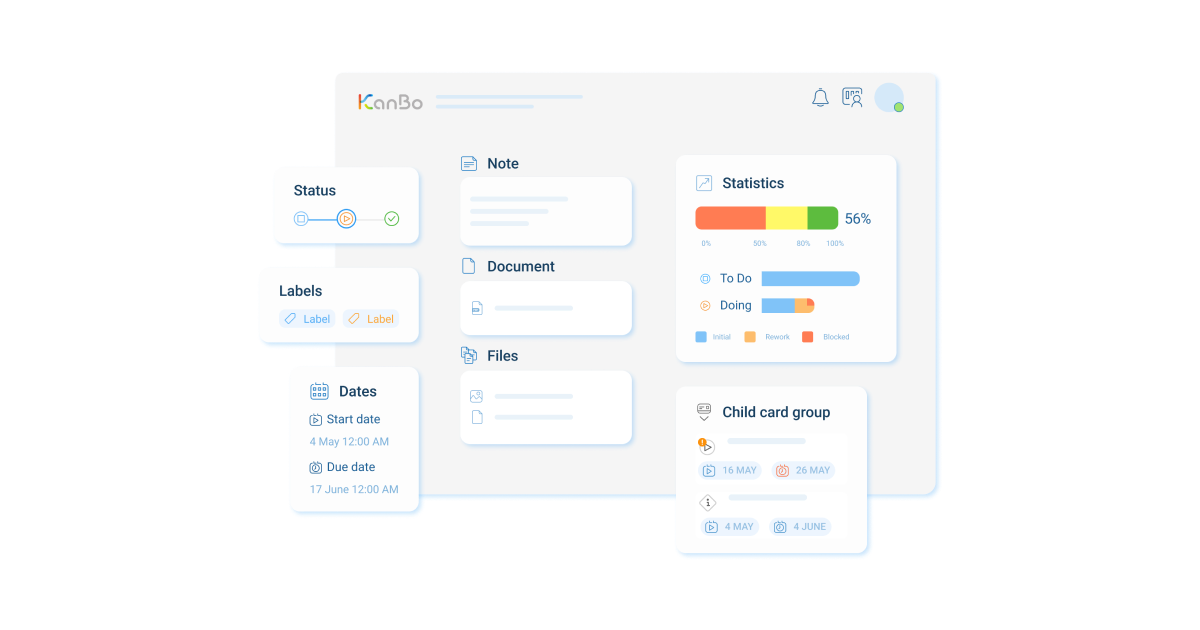
- Space: Create distinctive workspaces for respective teams, fostering clear communication and effective task management.
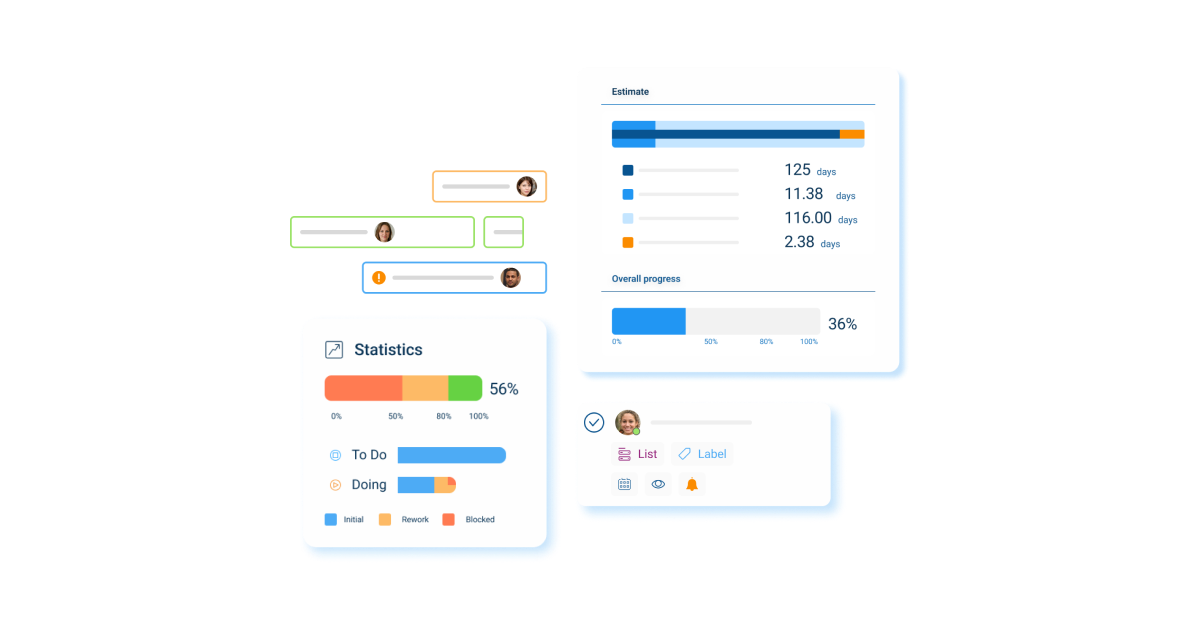
- Card: Utilize cards to map out various stages of trial design and analyses processes.
- Card Activity Stream: Monitor all activities on a card and foster transparency in the progression of tasks.

- Document Source: Centralize all essential documents related to the project, enhancing collaboration and reducing data fragmentation risk.

- User Activity Stream: Track individual contributions and performance, facilitating accountability and team synchronization.

- Calendar View: Plan and manage workload effectively, ensuring timely task completion.
What can you expect after solving this challenge?
Upon leveraging KanBo's features, you can expect to conquer these challenges and steer towards improved coordination among teams, accurately tracking the task progress. You can ensure that your teams always meet deadlines by planning and managing work effectively through the calendar view.
Not only can you foster transparency with effective tracking of individual and team activities, but also strengthen accountability. This will, in turn, boost morale and productivity within your teams, leading to improved and efficient clinical trial processes and deliverables.
In the long term, streamlined communication will cut down on wasted time and resources, speeding up decision-making processes. Tasks can be allocated and followed-up on more efficiently, ensuring that no detail is overlooked. As a result, the quality of your statistical deliverables will significantly improve, positively impacting your pharmaceutical projects.
In a nutshell, with KanBo, Senior Principal Biostatisticians can effectively collaborate within teams and ensure efficient and quality-controlled execution of pharmaceutical tasks and projects.

Table of Contents
KanBo – The Pharma-Focused Work Coordination Maestro
Experience ultimate task alignment, communication and collaboration
Trusted globally, KanBo, bridges the gap between management and engineering in complex pharmaceutical organizations. Seamless coordination, advanced project planning, and outstanding leadership are made possible through our versatile software. Stride toward your mission-critical goals with superior collaboration and communication.
KanBo for Senior Principal Biostatistician in the Pharmaceutical Industry: Boosting Collaboration, Efficiency and Quality
What do you need to know about this challenge?
A Senior Principal Biostatistician in the pharmaceutical industry plays a key role in driving the efficiency and efficacy of clinical trial teams and other quantitative partners. The challenge lays in coordinating with various teams to implement experimental designs and analyses, while ensuring timeliness and the quality of statistical deliverables. They have to maintain smooth communication flow, efficient task allocation, and effective progress tracking.
What can you do with KanBo to solve this challenge?
KanBo, with its array of features, provides a viable solution to these collaboration and management hurdles:
- Space: Create distinctive workspaces for respective teams, fostering clear communication and effective task management.

- Card: Utilize cards to map out various stages of trial design and analyses processes.

- Card Activity Stream: Monitor all activities on a card and foster transparency in the progression of tasks.

- Document Source: Centralize all essential documents related to the project, enhancing collaboration and reducing data fragmentation risk.

- User Activity Stream: Track individual contributions and performance, facilitating accountability and team synchronization.

- Calendar View: Plan and manage workload effectively, ensuring timely task completion.

What can you expect after solving this challenge?
Upon leveraging KanBo's features, you can expect to conquer these challenges and steer towards improved coordination among teams, accurately tracking the task progress. You can ensure that your teams always meet deadlines by planning and managing work effectively through the calendar view.
Not only can you foster transparency with effective tracking of individual and team activities, but also strengthen accountability. This will, in turn, boost morale and productivity within your teams, leading to improved and efficient clinical trial processes and deliverables.
In the long term, streamlined communication will cut down on wasted time and resources, speeding up decision-making processes. Tasks can be allocated and followed-up on more efficiently, ensuring that no detail is overlooked. As a result, the quality of your statistical deliverables will significantly improve, positively impacting your pharmaceutical projects.
In a nutshell, with KanBo, Senior Principal Biostatisticians can effectively collaborate within teams and ensure efficient and quality-controlled execution of pharmaceutical tasks and projects.

