Table of Contents
KanBo – The Pharma-Focused Work Coordination Maestro
Experience ultimate task alignment, communication and collaboration
Trusted globally, KanBo, bridges the gap between management and engineering in complex pharmaceutical organizations. Seamless coordination, advanced project planning, and outstanding leadership are made possible through our versatile software. Stride toward your mission-critical goals with superior collaboration and communication.
KanBo and the Global Regulatory Affairs CMC Manager: Streamlining Collaboration in Pharmaceutical Regulatory Processes
Understanding the Intricacies of Regulatory Collaboration
In the complex landscape of pharmaceuticals, a Global Regulatory Affairs CMC Manager navigates a web of intricate collaboration challenges. Ensuring harmonious alignment between diverse teams, change management processes, and multiple regulatory bodies is no small feat. Synchronizing global strategies, mitigating risks, and securing timely health authority approvals requires a platform that can handle multifaceted communication and project management with finesse.
What KanBo offers to Navigate Regulatory Collaboration
KanBo presents a suite of tools specifically designed to enhance coordination and strategic success in regulatory affairs:
- Strategic Roadmapping: Utilize KanBo's Timeline view to visualize regulatory milestones and create a clear, chronological path for CMC strategy execution, making it easier to anticipate key project phases and deadlines.
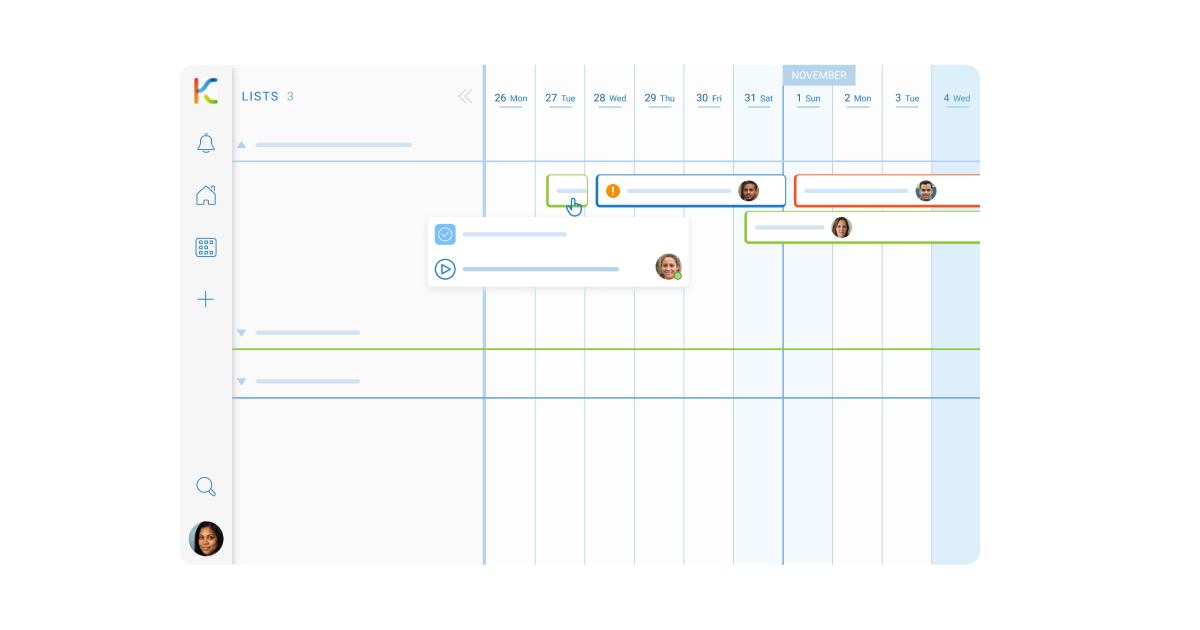
- Risk Mitigation Planning: Leverage the Card blocker feature to identify and categorize potential regulatory obstacles, ensuring teams can focus on proactive solutions and maintain momentum.
- Authority Interaction Logs: Employ the User activity stream to document interactions with health authorities, providing a detailed account of communications for reference and strategy refinement.

- Workflow Optimization: Take advantage of KanBo's Grouping feature to categorize regulatory affairs tasks by function or stage, streamlining process management and enhancing teamwork efficiency.

- Document Centralization: Improve access to relevant documents throughout the regulatory process by implementing Document source, thereby ensuring all team members have immediate access to critical information.

- Regulatory Update Alerts: Keep all stakeholders promptly informed with Notification, so any changes in the regulatory landscape can be swiftly incorporated into your strategies.

- Continuous Regulatory Education: Use the Card activity stream to track changes and updates to regulatory documents, enabling real-time education and ensuring the regulatory team is up to date.

The Long-term Advantages of Tackling Regulatory Collaboration with KanBo
Upon successfully leveraging KanBo to streamline regulatory collaboration, organizations can anticipate significant sustainable benefits. The heightened clarity and cohesion amongst teams lead to more efficient regulatory processes and reduced time to market for pharmaceutical products. There will be a noticeable decrease in compliance risks and enhanced adaptability to regulatory changes, thanks to the centralized platform and updated communication channels. Future enhancements in KanBo's feature set could potentially offer predictive analytics, further elevating the ability to preemptively address regulatory challenges and trends. As the regulatory landscape evolves, KanBo will remain an indispensable ally, consistently updating its offerings to meet the growing needs of Global Regulatory Affairs CMC Managers.

Table of Contents
KanBo – The Pharma-Focused Work Coordination Maestro
Experience ultimate task alignment, communication and collaboration
Trusted globally, KanBo, bridges the gap between management and engineering in complex pharmaceutical organizations. Seamless coordination, advanced project planning, and outstanding leadership are made possible through our versatile software. Stride toward your mission-critical goals with superior collaboration and communication.
KanBo and the Global Regulatory Affairs CMC Manager: Streamlining Collaboration in Pharmaceutical Regulatory Processes
Understanding the Intricacies of Regulatory Collaboration
In the complex landscape of pharmaceuticals, a Global Regulatory Affairs CMC Manager navigates a web of intricate collaboration challenges. Ensuring harmonious alignment between diverse teams, change management processes, and multiple regulatory bodies is no small feat. Synchronizing global strategies, mitigating risks, and securing timely health authority approvals requires a platform that can handle multifaceted communication and project management with finesse.
What KanBo offers to Navigate Regulatory Collaboration
KanBo presents a suite of tools specifically designed to enhance coordination and strategic success in regulatory affairs:
- Strategic Roadmapping: Utilize KanBo's Timeline view to visualize regulatory milestones and create a clear, chronological path for CMC strategy execution, making it easier to anticipate key project phases and deadlines.

- Risk Mitigation Planning: Leverage the Card blocker feature to identify and categorize potential regulatory obstacles, ensuring teams can focus on proactive solutions and maintain momentum.

- Authority Interaction Logs: Employ the User activity stream to document interactions with health authorities, providing a detailed account of communications for reference and strategy refinement.

- Workflow Optimization: Take advantage of KanBo's Grouping feature to categorize regulatory affairs tasks by function or stage, streamlining process management and enhancing teamwork efficiency.

- Document Centralization: Improve access to relevant documents throughout the regulatory process by implementing Document source, thereby ensuring all team members have immediate access to critical information.

- Regulatory Update Alerts: Keep all stakeholders promptly informed with Notification, so any changes in the regulatory landscape can be swiftly incorporated into your strategies.

- Continuous Regulatory Education: Use the Card activity stream to track changes and updates to regulatory documents, enabling real-time education and ensuring the regulatory team is up to date.

The Long-term Advantages of Tackling Regulatory Collaboration with KanBo
Upon successfully leveraging KanBo to streamline regulatory collaboration, organizations can anticipate significant sustainable benefits. The heightened clarity and cohesion amongst teams lead to more efficient regulatory processes and reduced time to market for pharmaceutical products. There will be a noticeable decrease in compliance risks and enhanced adaptability to regulatory changes, thanks to the centralized platform and updated communication channels. Future enhancements in KanBo's feature set could potentially offer predictive analytics, further elevating the ability to preemptively address regulatory challenges and trends. As the regulatory landscape evolves, KanBo will remain an indispensable ally, consistently updating its offerings to meet the growing needs of Global Regulatory Affairs CMC Managers.

